 
We include specific suggestions for how VSEPR-style rationalizations of molecular structure can be replaced with more accurate conceptions of hybridization and its relationship to electronegativity and molecular geometry, in accordance both with Bent's rule and the consistent features of modern wavefunctions as exhibited by natural bond orbital (NBO) analysis. VSEPR-style conceptions thereby detract from more accurate introductory-level teaching of orbital hybridization and bonding principles, while also requiring wasteful ''unlearning'' as the student progresses to higher levels. Using VSEPR theory, we predict that the two regions of electron density arrange themselves on opposite sides of the central atom with a bond angle of 180. VSEPRTheory(MolecularShapes) the central atom, X an atom bonded toA, E alonepair on A pairs on X or other atoms, but we dont care. Step 2: Determine the Central Atom of the Molecule. VSEPR-style representations of orbital shape and size are shown to be fundamentally inconsistent with numerous lines of experimental and theoretical evidence, including quantum mechanical ''symmetry'' principles that are sometimes invoked in their defense. total valence electron number in CO32- is 4 + 63 + 2 24. s orbitals are spherically symmetric around the nucleus - in each case, like a hollow ball made of rather chunky material with the nucleus at its centre.We describe the logical flaws, experimental contradictions, and unfortunate educational repercussions of common student misconceptions regarding the shapes and properties of lone pairs, inspired by overemphasis on ''valence shell electron pair repulsion'' (VSEPR) rationalizations in current freshmanlevel chemistry textbooks. The "s" tells you about the shape of the orbital. The "1" represents the fact that the orbital is in the energy level closest to the nucleus. 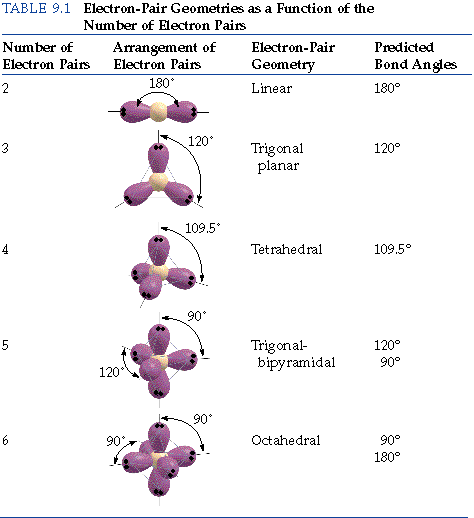
The orbital occupied by the hydrogen electron is called a 1s orbital. Our chemistry teacher told us that both VSEPR theory (which says that the electron pairs in the valence shell of an atom arrange themselves in such a way that repulsions among them are minimized and this arrangement of the electron pairs determines the shape of a particular molecule) and hybridization (which is the intermixing of a particular nu. You can think of an orbital as being the region of space in which the electron lives. VSEPR theory predicts the shapes of molecules, and hybrid orbital theory provides an explanation for how those shapes are formed. (b) Two of the hybrid orbitals on oxygen contain lone pairs, and the other two overlap with the 1 s orbitals of hydrogen atoms to form the OH bonds in H 2 O. BondingMOs have lower energy than the AO forming. It specifically uses the Coulombic repulsion between electrons as a basis for predicting electron arrangement. Such a region of space is called an orbital. (a) A water molecule has four regions of electron density, so VSEPR theory predicts a tetrahedral arrangement of hybrid orbitals. VSEPR theory (Valence Shell Electron Pair Repulsion theory) states that electrons repel each other because they are all negatively charged, so the bonds and lone pairs in a molecule will get as. Molecular Orbital (MO) Theory Total number of MOs formed, must be equal to that of AO combined to make them. The Valence-Shell-Electron-Pair-Repulsion (VSEPR) theory can predict molecular geometry by minimizing electron-electron repulsion. First, write the Lewis structure of the molecule, in this model, consider only the electron pairs of the central atom. The diagram shows a cross-section through this spherical space.ĩ5% of the time (or any other percentage you choose), the electron will be found within a fairly easily defined region of space quite close to the nucleus. Here are some guidelines for applying the VSEPR model: 1. In the hydrogen case, the electron can be found anywhere within a spherical space surrounding the nucleus. 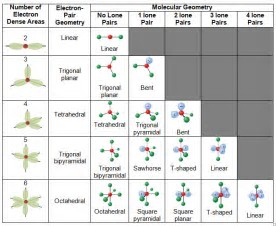
You keep on doing this over and over again, and gradually build up a sort of 3D map of the places that the electron is likely to be found. You have no idea how it got from the first place to the second. Soon afterwards, you do the same thing, and find that it is in a new position. Suppose you had a single hydrogen atom and at a particular instant plotted the position of the one electron. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
